
Introduction
Ammonia exposures don't just injure workers — they shut down facilities, generate millions in damages, and make headlines. The data backs this up: inorganic gases caused 52.9% of all acute occupational inhalation incidents investigated by OSHA between April 2011 and March 2022, with anhydrous ammonia accounting for 7.3% of those exposures.
The real-world consequences are severe. The 2024 Cuisine Solutions incident in Sterling, Virginia left 33 workers hospitalized, caused $3 million in property damage, and forced up to 38 days of facility shutdown — all from a gas used daily in refrigeration, agriculture, and manufacturing.
Ammonia is widely used, and widely mishandled. Effective safety depends on layered controls: proper PPE, disciplined handling procedures, adequate ventilation, gas detection systems, and comprehensive emergency preparedness working together as an integrated system.
TLDR
- Ammonia (NH₃) is a colorless, pungent gas; toxic and corrosive with serious exposure risks through inhalation, skin contact, or eye contact
- NIOSH recommends a 25 ppm TWA and 35 ppm STEL; OSHA enforces a 50 ppm PEL, with 300 ppm considered immediately life-threatening
- Wear ammonia-rated respirators, chemical-resistant gloves, and splash-proof goggles in ventilated areas with continuous gas monitoring
- Store in pressure-rated containers away from acids, halogens, and heat sources; never use copper, brass, or galvanized steel equipment
- In an emergency, evacuate immediately, flush eyes/skin for 15–30 minutes, and seek medical attention even if symptoms seem mild
Understanding Ammonia Hazards in the Workplace
Anhydrous ammonia (NH₃) is a colorless gas with a sharp, suffocating odor used extensively in refrigeration systems, agricultural fertilizers, and chemical manufacturing. It is shipped and stored as a liquefied compressed gas that boils at -28°F (-33.3°C).
Upon release, liquid ammonia expands at an approximately 850:1 ratio — one gallon of liquid produces 850 gallons of gas. Although ammonia vapor is technically lighter than air (vapor density 0.596), pressurized liquid releases form dense, two-phase aerosol clouds that slump to ground level and hug the terrain rather than dissipating upward.
Short-Term Health Effects
Ammonia is highly hygroscopic, meaning it absorbs moisture aggressively. Upon contact with human tissue, it reacts with moisture in the eyes, skin, and respiratory tract to form ammonium hydroxide, a corrosive alkaline solution that causes tissue liquefaction. Short-term exposure produces:
- Burning sensations in eyes, nose, and throat
- Severe coughing and shortness of breath
- Chemical burns to skin and eyes
- At concentrations above 300 ppm, pulmonary edema (fluid accumulation in the lungs)
Long-Term and Delayed Health Effects
Repeated low-level exposures cause cumulative damage that extends well beyond the initial incident. Long-term and delayed effects include:
- Chronic bronchitis and asthma-like symptoms from repeated low-level exposure
- Permanent lung tissue scarring from prolonged inhalation
- Permanent vision loss from extended eye contact
- Delayed-onset noncardiogenic pulmonary edema developing 12–72 hours after exposure — even in workers who show no immediate symptoms
This delayed pulmonary response makes post-incident medical observation mandatory for anyone involved in an ammonia release.
OSHA and Occupational Exposure Limits
Understanding regulatory thresholds is essential for making real-time safety decisions:
| Limit Type | Value | Meaning |
|---|---|---|
| OSHA PEL | 50 ppm (8-hour TWA) | Legal maximum average exposure over an 8-hour shift |
| NIOSH REL | 25 ppm (10-hour TWA) | Recommended safer limit for daily exposure |
| NIOSH STEL | 35 ppm (15-minute) | Maximum short-term exposure limit |
| IDLH | 300 ppm | Immediately Dangerous to Life or Health — requires SCBA |

The IDLH threshold of 300 ppm represents the concentration above which workers cannot escape without suffering permanent injury or death. At this level, air-purifying respirators fail, and only Self-Contained Breathing Apparatus (SCBA) provides adequate protection.
Fire and Explosion Risk
Ammonia is generally considered non-flammable under normal conditions, with a Lower Explosive Limit (LEL) of 15% and Upper Explosive Limit (UEL) of 28% by volume. However, at high concentrations in certain atmospheric conditions, it can ignite. The autoignition temperature is 1,204°F (650°C). At elevated temperatures above 840°F–930°F, ammonia decomposes into hydrogen and nitrogen gases, with hydrogen being highly flammable and adding significant explosion risk in fire scenarios.
The Olfactory Fatigue Trap
While ammonia's odor detection threshold is low (5–53 ppm), continuous exposure causes olfactory fatigue (adaptation). Workers exposed to low concentrations for extended periods lose the ability to detect escalating, life-threatening concentrations by smell alone. Odor alone is not a reliable warning system. Automated gas detection is the only dependable safeguard against this risk.
Ammonia Safety Guidelines
Effective ammonia safety requires layered controls that work together: engineering solutions (ventilation systems, gas detection), administrative practices (training programs, regular inspections), and individual protective measures (PPE use, procedure compliance). Skipping any layer creates gaps that increase injury risk, regulatory liability, and facility downtime.
General Safety Precautions
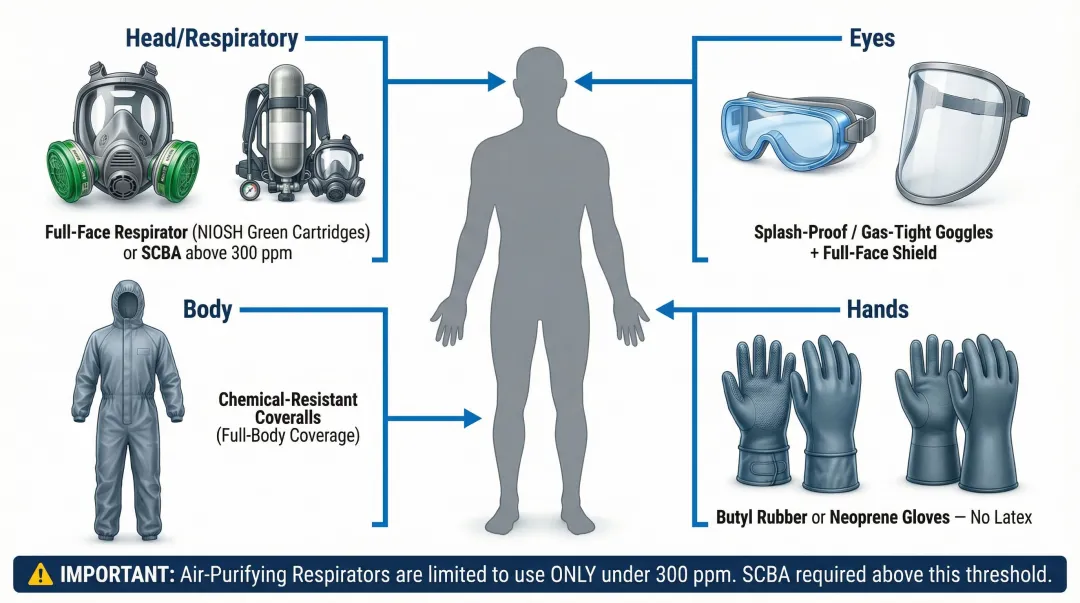
Personal Protective Equipment Requirements:
- Respiratory Protection: Full-face respirator with NIOSH-approved green ammonia/methylamine cartridges for concentrations up to 300 ppm; positive-pressure SCBA for concentrations above 300 ppm or unknown atmospheres
- Hand Protection: Chemical-resistant gloves made of butyl rubber, neoprene, or PTFE (Teflon)—never use latex gloves
- Eye Protection: Splash-proof goggles or gas-tight goggles; full-face shields when handling large quantities
- Body Protection: Chemical-resistant coveralls or suits for large-scale handling operations
Ventilation Requirements:
Workspaces must maintain continuous exhaust ventilation or local exhaust systems. ANSI/IIAR 2-2021 sets specific minimums for machinery rooms:
- Emergency ventilation rate: 30 air changes per hour (ACH)
- Exhaust fan discharge: Upward at minimum 2,500 feet per minute
Workers must never enter confined or poorly ventilated spaces without atmospheric testing and continuous monitoring. Natural ventilation alone is never sufficient in ammonia-risk areas.
Safe Storage and Handling
Proper Storage Practices:
- Use only pressure-rated, certified ammonia containers made of steel or ductile iron
- Store in cool, shaded areas to prevent pressure buildup from temperature increases
- Segregate from incompatible materials including acids, chlorine, halogens, and strong oxidizers
- Never use copper, brass, zinc, or galvanized steel equipment—ammonia rapidly corrodes these metals
- Label all storage areas with appropriate hazard markings
Hazard Identification and Regulatory Compliance:
Proper hazard identification—including pipe markers, warning signs, and container labels—is a regulatory expectation under OSHA's Hazard Communication Standard (29 CFR 1910.1200). Facilities storing 10,000 pounds or more of anhydrous ammonia trigger strict oversight under both OSHA's Process Safety Management (PSM) standard and EPA's Risk Management Program (RMP).
RE&D supplies durable safety signs, valve identification tags, pipe markers, and custom decals built for corrosive industrial environments—helping facilities meet these identification requirements.
Safe Handling Practices:
- Inspect all equipment (valves, tanks, hoses, pipelines) before each use for signs of corrosion, leaks, or damage
- Never transfer ammonia without pressure-rated equipment and proper training
- Follow lockout/tagout (LOTO) procedures during all maintenance activities
- Avoid working alone when handling large quantities
- Ensure emergency shutdown systems are accessible and clearly marked
Environmental and Workplace Conditions
Environmental conditions directly affect ammonia risk. High ambient temperatures raise vapor pressure in storage containers, increasing the likelihood of pressure relief valve discharge or catastrophic failure.
Poor airflow allows invisible gas accumulation in low-lying areas. During large releases, cold ammonia plumes can initially hug the ground before dispersing, creating concentrated exposure zones at floor level.
Gas Detection Systems:
Install fixed gas detection systems in all areas with ammonia risk. Configure detection systems to monitor critical thresholds:
- 150 ppm: ANSI/IIAR 2 requires automatic activation of visual indicators, audible alarms, and emergency exhaust ventilation
- 300 ppm: IDLH threshold—requires immediate evacuation and SCBA for entry
- 40,000 ppm (25% LEL): Automatic de-energization of compressors and pumps to prevent ignition sources

Alarm systems must trigger evacuation protocols before dangerous concentrations build. Never disable or ignore alarms to finish tasks.
Emergency Response for Ammonia Exposure
Emergency response must be pre-planned and regularly drilled. Workers in ammonia-risk environments should know the facility's Emergency Action Plan (EAP) and their specific role — whether to evacuate immediately or, if trained under HAZWOPER standards (29 CFR 1910.120), to actively respond. Improvising during a live release costs time and increases casualties.
Evacuation and Spill Containment
Evacuation Protocol:
- Immediately alert all personnel upon confirmed or suspected ammonia release using facility alarm systems
- Use pre-established evacuation routes that move workers upwind and away from the release plume
- Account for all personnel at designated assembly points located upwind and uphill from the facility
- Contact emergency responders immediately for releases exceeding incidental quantities
Recent Chemical Safety Board (CSB) investigations reveal that inadequate Emergency Action Plans often fail to account for wind direction, do not differentiate between indoor and outdoor releases, and lack clear shelter-in-place guidance. Ensure your EAP addresses these gaps.
Spill and Leak Containment:
- For small leaks (incidental releases), use ammonia-rated spill control kits if trained and properly equipped
- For large releases, do not attempt containment without HAZWOPER training and SCBA
- Isolate the area and defer to hazmat responders
- Audit all atmospheric relief discharge points to ensure they vent to safe locations away from building intakes, parking lots, and evacuation routes
First Aid Procedures
Inhalation Exposure:
- Move the affected person to fresh air immediately
- If breathing is labored or stopped, administer oxygen and call emergency services
- Do not re-enter the contaminated area without SCBA
Skin and Eye Contact:
- Flush affected skin or eyes with clean water for a minimum of 15–30 minutes
- Hold eyelids open during flushing to ensure thorough irrigation
- If clothing is frozen to skin by liquid ammonia, thaw with water before removing — pulling it off can tear tissue
- Remove contaminated clothing carefully and seal in plastic bags
- Seek immediate medical evaluation even if symptoms appear mild; effects can worsen hours after exposure

Critical Warning:
Symptoms of ammonia inhalation can be delayed. Pulmonary edema may develop 12 to 72 hours after exposure, making post-incident medical observation essential for anyone with confirmed or suspected inhalation. Never treat mild initial symptoms as a sign that someone is in the clear.
Common Ammonia Safety Mistakes to Avoid
Skipping Pre-Use Equipment Inspection:
Valves, hoses, and tanks degrade over time from corrosion and mechanical stress — skipping pre-use inspection is one of the leading causes of accidental ammonia release. A 2023 incident at Hilmar Cheese illustrates what's at stake: a worker cut into a live ammonia line because the task wasn't classified as permit-required, resulting in injury, system downtime, and a regulatory investigation.
Relying on Odor Alone as a Warning Signal:
While ammonia's smell is distinctive at low concentrations, olfactory fatigue occurs with prolonged low-level exposure. Workers underestimate accumulation because they can no longer smell it. Fixed gas detection equipment must be configured to activate emergency ventilation and trigger evacuation before dangerous concentrations build — smell alone cannot be trusted.
Inadequate PPE Selection or Fit:
Using the wrong respirator cartridge, loose-fitting goggles, or standard gloves instead of chemical-resistant ones dramatically reduces protection. Air-purifying respirators are limited to concentrations below 300 ppm — above that threshold, only SCBA provides adequate protection.
Inspect PPE for damage and confirm proper fit before every use, not just during initial training.
Conclusion
Ammonia safety depends on layered controls working together as an integrated system. PPE, proper storage practices, adequate ventilation, gas detection systems, durable hazard labeling, and emergency preparedness aren't independent checkboxes—each layer reinforces the others. Facilities that make ammonia safety part of daily operations, rather than an afterthought, protect workers, reduce downtime, and stay ahead of compliance requirements.
Put that commitment into practice by covering these four fundamentals:
- Train comprehensively — ensure every worker handling ammonia knows the hazards, controls, and emergency procedures
- Identify hazards clearly — maintain accurate, durable labeling and signage at every storage point and handling area
- Inspect equipment regularly — catch worn seals, faulty detectors, and damaged PPE before they become incidents
- Drill emergency response — run realistic scenarios so crews respond quickly and correctly when it counts
Getting these right consistently is far less costly—in time, money, and human risk—than recovering from a preventable incident.
Frequently Asked Questions
What are the safety precautions for ammonia?
Core precautions include wearing ammonia-rated respirators, chemical-resistant gloves, and splash-proof goggles; ensuring adequate ventilation with continuous gas monitoring; storing ammonia in pressure-rated steel containers away from incompatible materials; and maintaining pre-planned, regularly drilled emergency response procedures.
What is the OSHA standard for ammonia?
OSHA's Permissible Exposure Limit (PEL) for ammonia is 50 ppm as an 8-hour time-weighted average (TWA). NIOSH recommends stricter limits: 25 ppm TWA and 35 ppm STEL. OSHA's HazCom Standard (29 CFR 1910.1200) additionally governs labeling, Safety Data Sheets, and worker training.
What happens if you inhale a small amount of ammonia?
Low-level inhalation typically causes irritation of the nose, throat, and eyes, along with coughing. However, symptoms can worsen hours after exposure, and repeated low-level exposures cause cumulative respiratory damage including chronic bronchitis and permanent lung tissue scarring. Seek medical evaluation for any significant exposure.
What PPE is required when working with ammonia?
Required PPE includes:
- Respirator: Ammonia-rated full-face respirator with NIOSH-approved green cartridges; SCBA for concentrations above 300 ppm
- Hand/body protection: Chemical-resistant gloves (butyl rubber or neoprene), splash-proof goggles, and chemical-resistant coveralls
PPE selection must match the concentration level and task type.
Is ammonia flammable or explosive?
Ammonia is generally considered non-flammable at ambient conditions but can ignite at high concentrations (15%–28% by volume in air). At temperatures above 840°F–930°F, ammonia decomposes into highly flammable hydrogen gas, creating explosion risk in fire scenarios or near high-temperature equipment.
What are the first aid steps for ammonia skin or eye exposure?
Immediately flush skin or eyes with clean water for 15–30 minutes, holding eyelids open during eye irrigation. Remove contaminated clothing carefully (thaw frozen clothing with water before removal). Move the person to fresh air and seek immediate medical attention, as symptoms may worsen hours after the initial incident.